- No.101 Wanbo 1st Road, Nancun Town, Panyu District, Guangzhou, Guangdong, China
- 0086-020-84886013,84886093,84886091,84886012
- sales@finegosteel.com
- Complaints & Suggestions : suggest@finegosteel.com
Industry News
What Causes Hydrogen-Induced Cracking In LSAW JCOE Pipes For Hydrogen Transport?
You need safe pipelines for hydrogen. Sudden ruptures cause huge money losses. This invisible killer threatens your projects. I will show you how to stop this exact problem. Hydrogen-induced cracking in lsaw jcoe pipes for hydrogen transport happens when hydrogen atoms enter the steel. These atoms gather inside the metal. The internal pressure builds up. This pressure cracks the pipe. Bad manufacturing processes, wrong steel ingredients, or harsh working environments cause this dangerous failure.

You often think your steel pipes are perfectly safe after standard tests. Hidden dangers always lurk deep inside the metal structure. We will uncover the exact triggers. These triggers turn a strong pipe into a ticking time bomb. You must keep reading to save your future projects.
Where Does The Destructive Hydrogen Come From During Manufacturing?
Manufacturing traps bad hydrogen inside the metal. This hidden gas ruins the pipe strength. You must understand these exact sources. You need this knowledge to prevent pipe failures early. Destructive hydrogen enters steel during melting, rolling, and welding. The molten steel needs proper dehydrogenation. Unpurified steel keeps the hydrogen inside. Submerged arc welding uses wet flux or rusty wire. The welding heat turns this water into hydrogen. Fast cooling locks this gas inside the weld seam forever.
The Melting And Rolling Stage
Steel making requires high temperatures. Gases mix with the liquid metal. Factories must remove this gas. Bad factories leave hydrogen atoms inside the solid steel plate. These atoms are very small. They move easily through the tiny spaces inside the metal. Test data shows a high danger level. The hydrogen amount sometimes exceeds 2 parts per million (ppm). This small amount increases the cracking risk three times.
The Submerged Arc Welding Process
Welding is the most common way for hydrogen to enter lsaw jcoe pipes for hydrogen transport. The electric arc creates massive heat. This heat breaks down water from the air. Wet welding flux holds water. Dirty welding wires hold water. The high heat turns this water into hydrogen gas. The melted metal absorbs this gas. The weld joint cools down fast. The hydrogen cannot escape into the air. It locks inside the weld joint permanently. We at Finego Steel test our materials strictly. Bad factories use wet flux. The hydrogen level in their welds reaches 15 ppm. This high number fails all basic safety tests.
| Hydrogen Source | Entry Point | Danger Level |
|---|---|---|
| Unpurified Liquid Steel | Metal core | High |
| Wet Welding Flux | Weld seam | Very High |
| Dirty Welding Wire | Metal surface | Medium |
How Do The Material Characteristics Of Steel Increase Cracking Risks?
You buy very strong steel. The steel still cracks. Bad internal metal structures trap hydrogen easily. I will show you the exact chemical problems behind this brittle metal disaster. The chemical composition determines the hydrogen resistance. High carbon creates pearlite. Pearlite traps hydrogen atoms. High sulfur forms manganese sulfide impurities. These impurities create weak spots. Hydrogen gathers around these weak spots. The gas forms small cracks. These small cracks destroy the pipe structure from the inside.
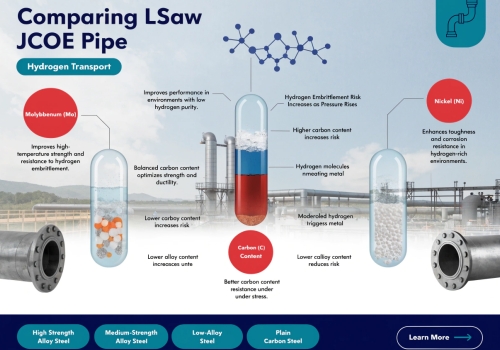
The Danger Of Carbon And Sulfur
Different steel types react differently to hydrogen. The basic ingredients change everything. High carbon levels create a material called pearlite. Pearlite meets ferrite inside the steel. This meeting point grabs hydrogen atoms. The atoms gather and push against the metal. The metal breaks. Sulfur acts like a slow poison. Sulfur mixes with manganese. This mix forms weak spots inside the steel. These spots do not hold tightly to the surrounding metal. Hydrogen loves to hide around these weak spots. Laboratory tests show a scary fact. The sulfur level goes from 0.01% to 0.03%. The steel becomes very brittle immediately.
The Problem With Microstructures
High-strength metals like X70 and X80 have complex insides. Bad heat treatment makes hard shapes. Engineers call these shapes martensite. These shapes have too much internal stress. They act like strong cages for hydrogen. The hydrogen enters the cage. The hydrogen cannot leave the cage. The pressure grows. The pipe eventually breaks. Tiny holes in the steel also act as starting points. Cracks start at these tiny holes. The cracks grow very fast.
| Chemical Element | Internal Result | Impact On Cracking |
|---|---|---|
| High Carbon | Forms pearlite | Traps hydrogen atoms |
| High Sulfur | Forms manganese sulfide | Creates weak spots |
| Bad Heat Treatment | Forms martensite | Increases internal stress |
What Production Process Flaws Lead To Hydrogen-Induced Cracking?
Pipe shaping and pipe heating create severe internal stress. This stress works with hydrogen to destroy pipes. We must control every production step to stop these expensive field failures. Poor JCOE forming leaves high residual stress in the steel. Incorrect welding energy changes the metal grain size. Bad factories skip the post-weld heat treatment. This skipping leaves hydrogen trapped inside. Wrong tempering temperatures fail to fix the brittle structures. This failure ruins the hydrogen cracking resistance completely.
The Stress From JCOE Forming
Making a round pipe from a flat plate requires huge force. The JCOE method bends the steel step by step. The machine bends the steel too fast. The machine bends the steel unevenly. The steel holds tension inside. We call this tension residual stress. The metal wants to spring back. The metal cannot spring back. This stress helps hydrogen atoms break the steel. Research gives us a clear warning. The internal stress reaches 60% of the metal limit. The cracking chance goes up five times. We at Finego Steel strictly control this bending process.
Welding And Heating Mistakes
The welding machine uses heavy electrical energy. Too much energy makes the metal grains too big. Big grains weaken the pipe. Too little energy cools the weld too fast. Fast cooling makes the metal hard. Fast cooling makes the metal brittle. Factories must bake the pipe after welding. They keep the pipe at 200 to 300 degrees Celsius for two hours. This baking pushes the hydrogen out. Bad factories skip this step to save time. This skipping ruins lsaw jcoe pipes for hydrogen transport. Wrong tempering temperatures ruin the metal structure completely. The hydrogen cracking test pass rate drops heavily.
| Process Flaw | Physical Effect | Failure Risk |
|---|---|---|
| Uneven Steel Bending | High residual stress | Increases risk 5 times |
| Skipping Heat Baking | Traps hydrogen in weld | Very High |
| Wrong Tempering Temp | Leaves brittle structure | Test pass rate drops |
How Do External Environmental Conditions Accelerate The Cracking Process?
Perfect pipes still break in bad working environments. Acid gases and high pressure speed up the cracking. You must match the pipe quality to your harsh working conditions. Harsh environments accelerate hydrogen cracking. Hydrogen sulfide gas creates new hydrogen atoms. These atoms enter the metal. High temperatures help these atoms move faster. High pressure forces more hydrogen into the steel. Repeated pressure changes cause fatigue. This fatigue makes small hydrogen cracks grow rapidly.
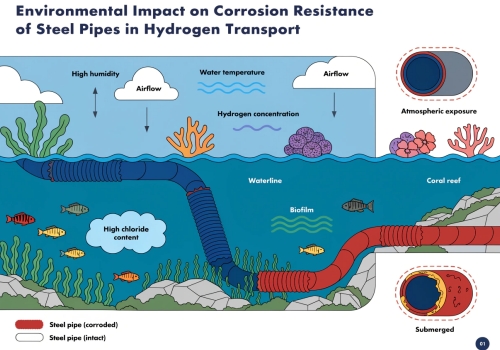
The Threat Of Hydrogen Sulfide
A perfect pipe leaves the factory. The working environment tests the pipe immediately. Oil fields and gas fields contain acid gases. Hydrogen sulfide is the worst enemy. This bad gas eats the metal surface. This eating process makes new hydrogen atoms. The gas helps these atoms push into the steel. The gas breaks the natural protective skin of the steel. This break makes the corrosion happen much faster. The hydrogen sulfide reaches 0.01% of the total gas volume. The cracking danger explodes immediately.
Temperature And Pressure Effects
Temperature changes everything. Hot environments give hydrogen atoms more energy. The atoms run fast inside the metal. High pressure pushes even more gas into the pipe walls. Deep sea pipes face terrible conditions. The deep sea is cold. The deep sea has high pressure. Cold water makes the steel brittle. High water pressure pushes the hydrogen inside. We call this a double risk. The pipe pump turns on. The pipe pump turns off. This action creates changing stress. This changing stress makes tiny cracks grow big very fast.
| Environmental Factor | Action | Result |
|---|---|---|
| Hydrogen Sulfide | Eats protective layer | Forces hydrogen into steel |
| High Temperature | Energizes atoms | Speeds up atom movement |
| High Pressure | Squeezes metal pipe | Pushes more gas inside |
Conclusion
Hydrogen-induced cracking destroys pipes through bad steel, poor manufacturing, and harsh environments. You can prevent this failure. You must choose reliable suppliers and control every production step strictly.
- Prev : How Do We Manufacture a High-Quality Cold Rolled Seamless Tube?
- Next : No matching information found

 Language
Language




